Catechol oxidaseCatechol oxidase is the enzyme responsible for the browning of fruit. It is easy to prepare from a number of different sources – bananas are particularly good – and the reaction is readily followed using a colorimeter.
Catechol oxidase has a number of alternative names, (Polyphenol oxidase,
Diphenol oxidase, Tyrosinase, etc,
The reaction catalysed is the oxidation of catechol to the yellow product 1,2-benzoquinone.
catechol + ½O2 → 1,2-benzoquinone +
H2O
On exposure to air there is a further reaction in which the yellow benzoquinone is converted to dark brown melanin. This reaction can be continuously monitored with the colorimeter and is particularly suitable for investigations into the effect of pH and the effects of competitive and non-competitive inhibitors. Dopa oxidase is a similar phenol oxidase and a good alternative to this assay
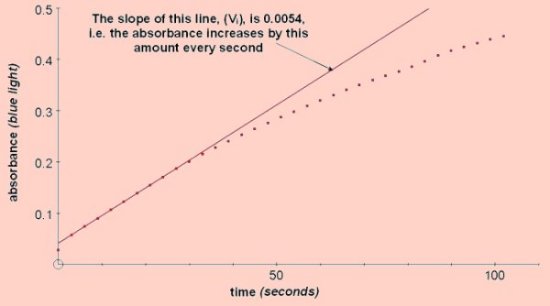
Absorbance vs Time
Effect of catechol concentration 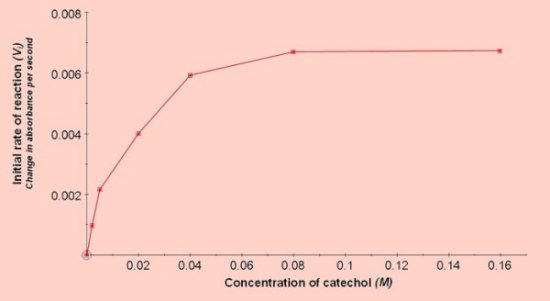 Effect of pH (results of 2 experiments) 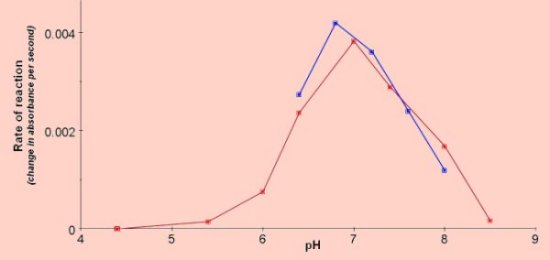 Effect of competitive and non-competitive inhibitors 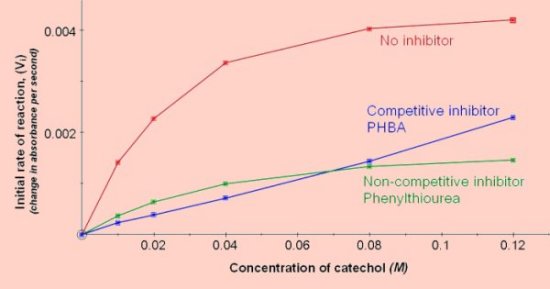
Enzyme extraction
To prepare the enzyme blend banana with two volumes of water or just squash the banana with a fork and crush it in a pestle and mortar with two volumes of water. 20g of banana with 40cm3 of water will give plenty of extract for most purposes. Filter the extract through several layers of butter muslin and store refrigerated. Reaction mixture
The reaction can be performed at room temperature, (results reported here were obtained at 25°C). The optimum pH is around 6.8 and catechol concentrations between 0.01M and 0.05M (final concentration in the cuvette) should give a good linear response when the concentration is plotted against the rate of reaction. Catechol MWt = 110Investigating the effect of pH
A reaction mix containing 0.01M catechol should give a good response at 25°C. (0.3mls 0.1M catechol + 2.6mls buffer added to 0.1mls enzyme extract in a standard cuvette). The initial straight line portion of the graph of absorbance against time gives Vi – the initial rate of reaction. Inhibitors
0.1cm3 of a 1M stock solution (dissolved in ethanol) added to a 3cm3 reaction mix is a suitable concentration to demonstrate inhibition.
0.1cm3 of a 0.02M stock solution added to a 3cm3 reaction mix is a suitable concentration.
|