PepsinThe slow breakdown of albumen proteins gives good results with continuous colorimetry and is well suited for the study of the effect of pH on enzyme activity. Pepsin (EC 3.4.23.1) is a proteolyic (protein splitting) enzyme produced in the gastric glands lining the stomach of vertebrates. It is produced as pepsinogen, a larger molecule that is activated by hydrochloric acid to the smaller pepsin molecule. Pepsin can only work in a low pH environment, (optimum pH~1.8) Pepsin does not break the bonds between every amino acid so the products of the reaction are polypeptides of varying lengths. Pepsin can conveniently be assayed using a cloudy suspension of egg white (albumen), which will slowly clear as the albumen protein is broken down into small, soluble polypeptides. The preparation of a suitable suspension is critical for the success of this assay (see Methods).
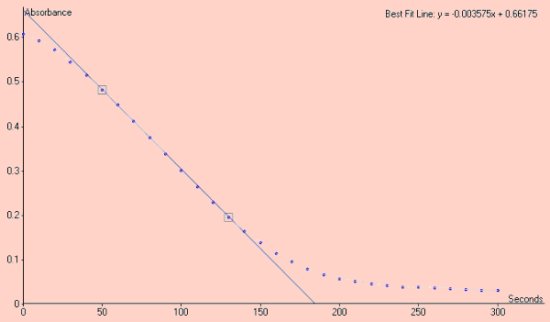
Absorbance vs Time
The rate of the reaction shown below is the slope of the line in the region between absorbance 0.5 and 0.2, given as
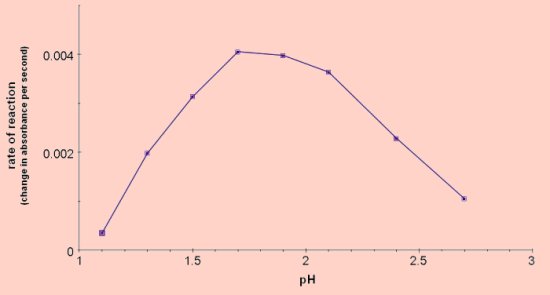
Effect of pH on reaction rate
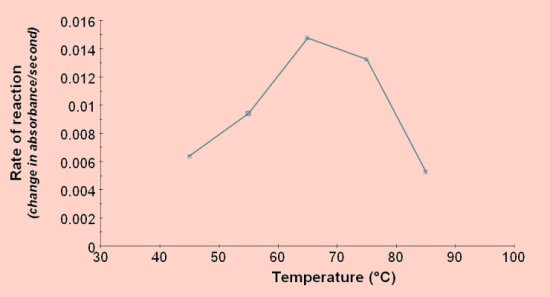
Effect of temperature on reaction rate
Warm both solutions to the reaction temperature before mixing the egg white suspension with the enzyme. The pH of the egg white suspension should be adjusted to pH 1.8 after it has reached the required temperature.
To prepare an suspension of egg white
You should be left with an opaque suspension. This can be diluted 1:1 with water or buffer for use This is a rather unpredictable outcome. Different eggs give different suspensions and if the suspension is not sufficiently opaque, with an absorbance greater than about 0.7, the results are likely to be disappointing. Reducing the ratio of water to egg white will probably help. Adjusting the pH
The egg white suspension can be acidified as it is, or diluted 1:1 with water first.
To investigate the optimum pH a range of different solutions can be set up as indicated in the table. These are only valid at To 50cm3 of a 1:1 mixture of egg white suspension and water add 2M HCl according to the table below.
 Click to download a pdf file containing much of the information about this
topic.
Click to download a pdf file containing much of the information about this
topic.
|