Beer's LawBeer’s Law (or Beer-Lambert, Lambert-Beer or Beer-Lambert-Bouger) states that there is a logarithmic relationship between the transmission of light through a sample and the product of the absorption coefficient of the absorbing substance times the distance of the light path through the sample. In practice this means that there is a directly proportional straight line relationship between concentration and absorbance. There are certain conditions that must be met for the law to apply. The most important of these in the context of colorimetry is that the incident light should be as near monochromatic as possible, or at least should have be equally absorbed throughout its range of wavelengths. If this condition is not met the relationship will be non-linear.
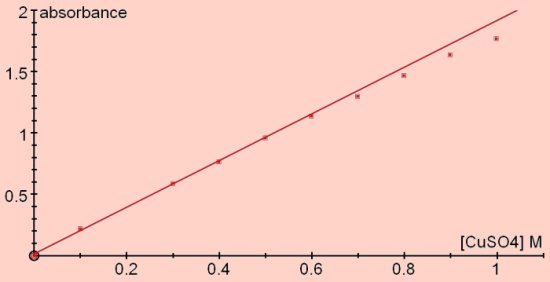
In general colorimeters, whether they use LEDs or coloured filters to create their light beam, do not have the narrow wavebands that can be achieved with spectrophotometers. So precise linear relationships are not always achievable and depend on a good match between the incident waveband and the absorbed waveband. Absorbance (red light) versus concentration (M) of copper sulphate
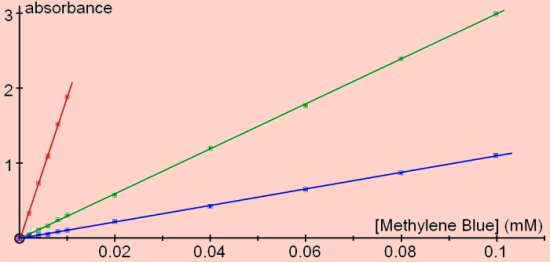
Absorbance versus concentration of methylene blue
Note that the colorimeter display will not show values of absorbance higher than 1.999. Higher values can be displayed with the software or by converting the transmittance values (0.1% transmittance = an absorbance of 3)
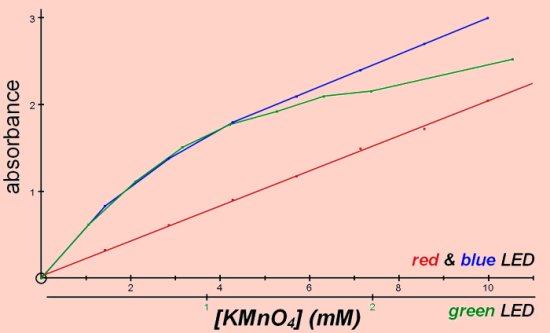
Absorbance versus concentration of potassium (VII) manganate, (potassium permanganate).
The curves with blue and green are a result of unequal absorbance of the emitted wavelengths. The shapes of the curve differ because the inequality across the wavelength range is not the same with both LEDs. The green LED results have been rescaled for comparison, potassium permanganate absorbs green light far more readily than red or blue.
|